Imagine the sound of rain tapping against a window pane. A grandchild's secret whispered in your ear. The satisfying crunch of autumn leaves underfoot. These aren't just noises; they are the textures of life, the subtle brushstrokes that paint your reality. What if those colors began to fade, slowly, almost imperceptibly, leaving the world a little less vibrant? This guide isn't just about hearing more; it's about feeling more. And the journey back to that vibrant world begins with three critical letters: FDA.
That seal on FDA approved hearing aids is more than a label. It’s a promise. A promise of safety, of proven effectiveness, and of a bridge back to the moments you've been missing.

From a Muffled World to a Life in High Definition
Does the world sometimes feel like a radio station that's just out of tune? You lean in, strain to catch the melody of a conversation, but the static of background noise wins. The TV volume creeps up, notch by notch. You find yourself nodding and smiling, hoping you caught the punchline of a joke you couldn't quite hear. It's a quiet retreat, a slow-motion withdrawal from the vibrant tapestry of sound.
This slow fade can be profoundly isolating. It’s the missed connection in a crowded room, the lost intimacy in a quiet conversation. A creeping suspicion begins to form in the back of your mind: Is this just how it is now?
I'm here to tell you it's not. What if you could flip a switch? What if you could dial the clarity of your life back up, restoring the sharp edges to conversations and the rich bass to your favorite songs?
A Secret Map Back to the Sounds You've Lost
Forget everything you think you know about hearing aids. This isn't a dense technical manual filled with jargon. Think of this as your personal map, drawn by someone who knows the terrain, leading you straight back to the world of sound you thought was gone forever. We're going to cut through the confusion, bypass the intimidating price tags, and focus on the one landmark that truly matters.
That landmark is the FDA's seal of approval. This simple designation is the bright, clear line separating a genuine medical device from a cheap, ineffective amplifier. It's your assurance that the device has been rigorously evaluated against strict government standards for:
- Safety: You can trust it won't harm your ears. It’s designed for your well-being.
- Effectiveness: It's been proven to do what it claims—help you hear better.
- Honesty: The information you get is clear, accurate, and free of misleading claims.
This is not a trivial detail. For the nearly two-thirds of Americans aged 70 and older living with hearing loss, understanding this seal is the first, most crucial step toward regaining control. It's the moment you move from a place of uncertainty to a position of power.
This journey is about more than just volume. It's about reclaiming your seat at the dinner table. It's about feeling the emotional swell of an orchestra. It's about rediscovering the unshakable confidence that comes from catching every single word. The path begins here, and that FDA seal is the North Star guiding your way.
What Does the FDA Seal Actually Promise Your Ears?
That phrase, "FDA Approved," feels solid, doesn't it? It resonates with trust. But what's really happening behind the scenes before that promise is attached to a sensitive device you'll wear every day?

Imagine a team of meticulous inspectors, scientists, and engineers. Their entire focus is on three non-negotiable questions: Is this device safe for you? Does it actually work as promised? Are the claims made about it true? This isn't just about turning up the volume; it's about certifying a piece of technology as a genuine health tool that can change your life without putting you at risk.
This is where medical science and consumer protection intersect, creating a foundation of evidence and accountability you can build your confidence on.
The Different Roads to FDA Approval
Not all hearing aids travel the same path to get the FDA's blessing. Just like a driver needs a special license for a commercial truck versus a family car, the FDA tailors its scrutiny to the device's complexity and purpose. This tiered system is designed for one thing: your safety.
For decades, the main road was reserved for prescription hearing aids. These are Class I or Class II medical devices, and getting one traditionally required a visit to an audiologist. The FDA’s review, often through a process called a "510(k) clearance," meticulously verifies that a new device is every bit as safe and effective as devices already proven and on the market.
But then, a revolutionary new highway was built.
A New Dawn of Access: The OTC Revolution
Imagine the frustration of needing reading glasses but being told you had to get a prescription and pay a specialist thousands of dollars. It sounds absurd, right? For years, that was the reality for hearing loss. The creation of the Over-the-Counter (OTC) category for hearing aids changed everything.
This wasn't about lowering standards; it was about re-engineering them for a new era of consumer empowerment.
The objective was crystal clear: create a safe, effective, and accessible pathway for millions of adults with mild-to-moderate hearing loss, removing the unnecessary gatekeepers. The FDA established a new, robust set of rules governing everything from maximum sound output to clarity of instructions.
To sell an OTC hearing aid, a manufacturer must register their company and list their device with the FDA, certifying that it meets every single one of these stringent requirements. It’s a system built on trust and accountability, designed to help you make a smart, informed choice. Our guide on how to buy hearing aids explores how to navigate these exciting new options.
What That Seal Ultimately Guarantees You
So when you choose an FDA-compliant hearing aid, what is the core promise you receive? You're guaranteed a device that has passed through a gauntlet of federal regulations designed solely for your protection.
- Verified Safety: The device is built to operate within safe acoustic limits, ensuring it won't damage your hearing further.
- Proven Performance: It must amplify sound in a way that is genuinely beneficial for individuals with hearing loss.
- Unquestionable Clarity: The packaging must tell you exactly who the device is for, how to use it safely, and list any critical warnings.
Understanding this framework is essential. The FDA’s oversight is your ultimate benchmark of quality, your assurance that the product you're choosing is a legitimate medical device—not a cheap amplifier—and your safest first step on the path back to the sounds you love.
The Day the Walls Came Tumbling Down
For generations, the path to better hearing was a fortress, protected by high walls of cost, complexity, and gatekeepers. It was a frustrating maze of appointments, fittings, and a final price tag that locked millions of people out.
But what if someone just decided to tear those walls down? What if you could access help for your hearing with the same ease as buying reading glasses at a corner store?
A quiet revolution was building, not in a research lab, but in the halls of government. It was sparked by a single, powerful idea: you should have the power to take the first step yourself. This was the birth of the Over-the-Counter (OTC) hearing aid—a fundamental shift in philosophy designed to put control back into your hands.
From a Visionary Idea to a New American Reality
This movement was driven by leaders who saw a system that was failing the very people it was meant to serve. They recognized that the high costs and logistical hurdles were creating a silent epidemic, leaving nearly two-thirds of Americans aged 70 and older struggling alone.
Their relentless work resulted in a historic law that directed the FDA to create a brand-new category of FDA approved hearing aids. The mission was to maintain the highest standards of safety while finally removing the prescription barrier for adults with perceived mild to moderate hearing loss.
The entire landscape of hearing health changed forever on October 17, 2022. On that day, the FDA's new rule went into effect, and for the first time in history, over-the-counter hearing aids appeared on store shelves. You can explore the full history of this important regulatory shift to grasp the monumental impact of this moment.
Who Is This Revolution For?
With this new freedom came a critical question: who is this designed to help? The FDA drew a clear and specific line to ensure these devices would be used safely and effectively.
OTC hearing aids are built for adults aged 18 and older who perceive they have mild to moderate hearing loss. They are not intended for children or for individuals with severe or profound hearing impairment.
Think of it as the difference between self-treating a headache with aspirin versus seeing a neurologist for chronic migraines. OTC devices are for when you first notice the world's sharp edges are starting to soften.
- You find yourself turning up the volume on the TV or phone, just a little higher than everyone else.
- You consistently ask people to repeat themselves, especially in a group.
- You lose the thread of conversation in a noisy restaurant or at a family gathering.
If these scenes feel familiar, you are the person this revolution was built for.
A New Universe of Possibility and Innovation
The arrival of OTC hearing aids didn't just add a new product to the market; it ignited a firestorm of innovation. By opening the floodgates, it challenged companies to engineer better, more user-friendly, and more affordable solutions that could be delivered directly to your door.
This means you are no longer trapped between the choice of struggling in silence and spending a small fortune. Brands like HearDirectClub can now deliver FDA-registered Class I devices directly to you, pre-programmed for the most common hearing loss profiles and ready to use in minutes.
The result is the democratization of hearing health. The power has shifted, giving you direct access to safe, life-changing technology. This is the moment hearing health finally belongs to everyone.
Which Path to Clarity Is Yours to Take?
You now stand at a pivotal intersection on your journey to better hearing. One path leads to the accessible, self-directed world of Over-the-Counter (OTC) hearing aids. The other follows the traditionally guided route of prescription devices.
Which path is yours? Let’s demystify the choice.
Think of an OTC hearing aid like a world-class, ready-to-wear suit. It’s crafted with expert design to fit most people exceptionally well, offering superb quality right off the rack without the time and expense of a bespoke tailor. A prescription device is that bespoke suit—measured, cut, and stitched by a master tailor to fit your unique form, and only yours.
Both are excellent choices. The right one simply depends on your individual needs.
First, You Must Understand Your Own Map
The journey begins with an honest assessment of where you are right now. Do you find yourself just beginning to struggle in noisy restaurants? Do you ask your family to speak up more often? This typically signals the mild to moderate hearing loss that FDA approved hearing aids in the OTC category are masterfully designed to address.
However, if your hearing loss feels more significant, or if it’s accompanied by other signals like ringing in your ears (tinnitus), dizziness, or pain, then the personalized diagnosis and care of a hearing professional is the wisest and safest route.
This decision tree helps you see the choice clearly.
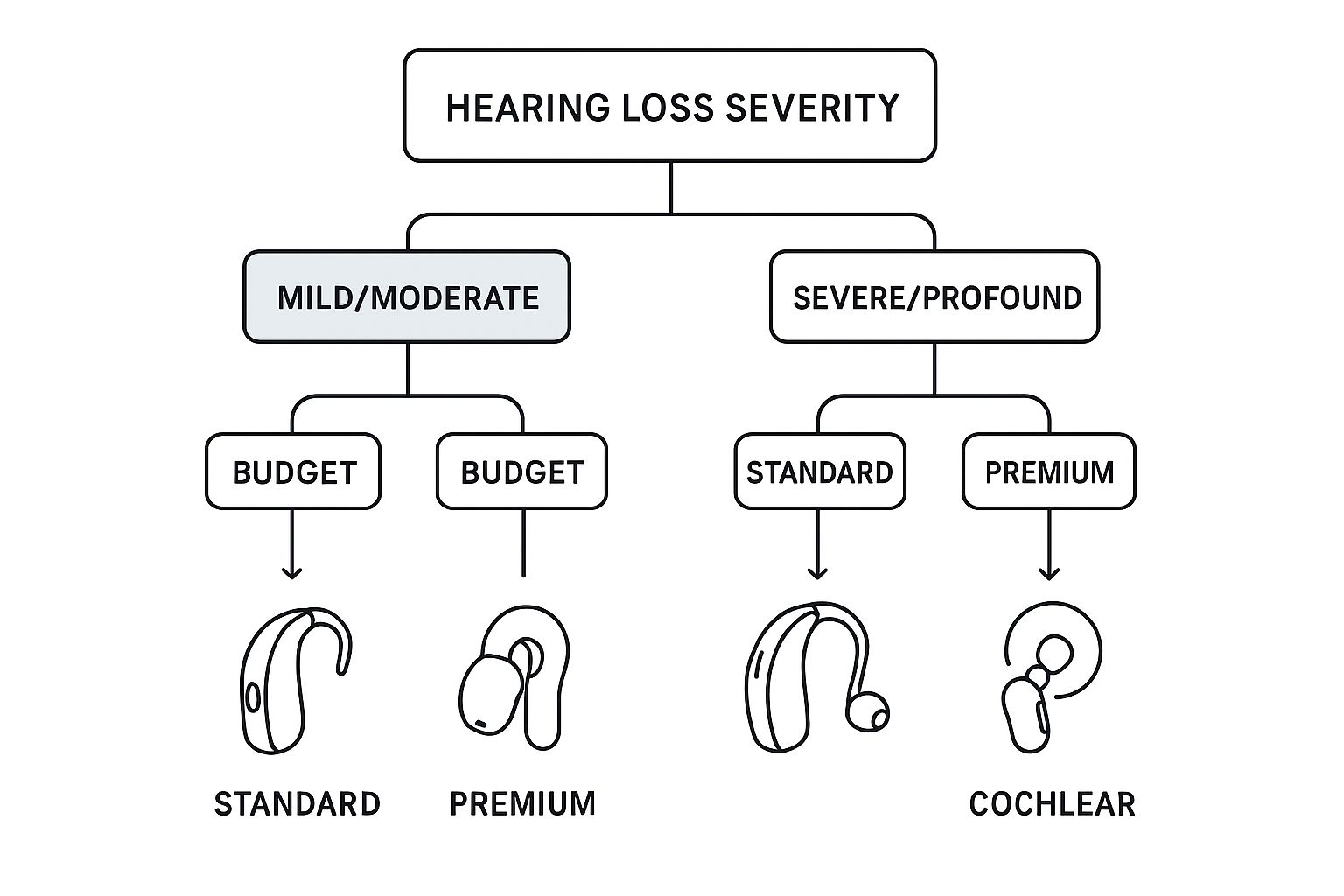
As you can see, for mild to moderate loss, OTC options provide a powerful and accessible solution. For anything more severe, the expertise of a professional and the precision of a prescription device are essential.
OTC vs. Prescription: A Clear-Cut Comparison
Let’s place these two powerful options side-by-side. Seeing the core differences will empower you to choose the path that will bring you success and satisfaction for years to come.
| Feature | OTC Hearing Aids | Prescription Hearing Aids |
|---|---|---|
| Accessibility | Purchase directly online or in-store, no prescription needed. | Requires a hearing test and fitting from an audiologist or specialist. |
| Ideal User | Adults (18+) with perceived mild to moderate hearing loss. | Anyone with any degree of hearing loss, from mild to profound. |
| Customization | Pre-programmed for common hearing loss patterns; some offer self-tuning. | Custom-programmed by a professional to your exact hearing profile. |
| Support System | Relies on manufacturer support (like our 24/7 HearExperts). | Involves ongoing professional appointments for adjustments. |
| Investment | Far more affordable, often between $300 and $600 per device. | A significant investment, typically ranging from $2,000 to $7,000 per pair. |
The choice crystallizes around a central theme: OTC offers independence, immediacy, and affordability. Prescription offers deep personalization and clinical oversight. There is no "best" answer—only what is best for you. For a deeper exploration, our guide on how to choose hearing aids unpacks even more personal scenarios.
See Yourself in These Stories
Let's make this real. Imagine two people, each starting their own journey.
-
Meet Sarah, the Social Butterfly: Sarah loves her weekly dinner with friends, but lately, the chatter of the restaurant drowns out the conversation at her own table. She's tech-savvy and wants a simple, powerful solution she can control herself, without the hassle of appointments. For Sarah, an FDA-registered OTC device from HearDirectClub is the perfect answer. It empowers her to solve her problem on her own terms.
-
Meet David, the Musician: David has noticed a sharp decline in his hearing in one ear, and certain musical frequencies sound distorted. His situation is more complex. David needs the diagnostic insight of an audiologist to uncover any underlying issues and to program a device with surgical precision. For him, a prescription device is the only path forward.
Which story resonates more with you? Answering that question is the key to unlocking your path back to a world of brilliant, beautiful sound.
How a Century of Genius Now Fits in Your Ear
That tiny, nearly invisible device sitting in the palm of your hand is not just a piece of plastic and wires. It's a miracle of miniaturization, the culmination of a century of relentless human ingenuity. The journey began with clumsy ear trumpets and body-worn battery packs the size of a book. Understanding this evolution isn't just a history lesson; it's appreciating the sheer genius that now lets you reclaim your hearing.
Imagine a time when "hearing help" meant strapping a giant horn to a box you had to carry everywhere. That was the era of analog. It made sounds louder, yes, but it amplified everything—the intimate whisper and the clattering dishes received the same brute-force boost. There was no intelligence, no finesse.
But deep inside research labs, a paradigm shift was occurring. The true revolution wasn't about making hearing aids smaller; it was about making them smarter.
The Quantum Leap to Digital Clarity
The seismic shift from analog to digital was like moving from a flickering candle to a modern LED bulb. The invention of the digital signal processing (DSP) chip gave the hearing aid something it never had before: a brain.
For the first time in history, a device worn in the ear could actively analyze sound. It could distinguish the rhythmic, predictable patterns of human speech from the chaotic randomness of background noise. This was the precise moment hearing aids evolved from simple amplifiers into sophisticated instruments of clarity.
A pivotal breakthrough occurred in the late 1980s. The first commercial all-digital hearing aid was developed in 1987, and by 1989, the first digital behind-the-ear model was transforming lives. This was supercharged when Widex launched the Senso, a device that could automatically adapt to its surroundings, filtering out noise in real-time. It forever changed the definition of what a hearing aid could do. You can explore the fascinating details of this technological leap and the history of hearing aids here.
That single innovation—the DSP chip—is the ancestor of every advanced feature we rely on today. Without it, there would be no feedback cancellation, no automatic environment detection, and no personalized sound profiles.
From a Smart Device to a Seamless Life
Once hearing aids had a brain, the next quest was to teach them how to communicate with the world. The arrival of wireless technology was the second great leap, transforming hearing aids from isolated sound processors into integrated personal communication hubs.
The introduction of Bluetooth connectivity, in particular, was a revelation. Suddenly, your hearing aid was no longer just for the room you were in. It could:
- Stream Phone Calls Directly: Conversations beamed with crystal clarity directly into your ears, hands-free.
- Become Your Personal Headphones: Music, podcasts, and television audio streamed in rich, high-fidelity sound.
- Enable Remote Care: Professionals could suddenly fine-tune your devices from hundreds of miles away, ushering in a new era of convenience.
This evolution turned the hearing aid from a medical necessity into a piece of multi-functional technology that integrates seamlessly into your life. Every feature in a modern FDA approved hearing aid—from all-day rechargeable batteries to the advanced tinnitus masking found in HearDirectClub devices—stands on the shoulders of these giants of innovation.
Your Final Questions, Answered with Clarity
Embarking on this journey can feel like learning a new language. It's natural to have questions. This final section is dedicated to cutting through the last remnants of fog, giving you the clear, direct answers you need to step forward with absolute confidence.
Consider this your personal FAQ for navigating the world of FDA approved hearing aids.
Are All Hearing Aids Sold in the US Governed by the FDA?
Yes, and this is the most important consumer protection you have. To be legally marketed as a "hearing aid" in the United States, a device must comply with FDA regulations. This applies to both traditional prescription devices and the newer Over-the-Counter (OTC) category.
However, a word of caution: be wary of "personal sound amplification products" (PSAPs). While they may look similar, PSAPs are not intended to correct hearing loss and are not regulated by the FDA as medical devices. Our guide on hearing aids versus hearing amplifiers breaks down the critical differences. Always confirm you are buying a true, FDA-compliant hearing aid.
How Do I Know If an OTC Hearing Aid Is the Right First Step for Me?
OTC hearing aids were created with a very specific person in mind: adults aged 18 and older who perceive they have mild to moderate hearing loss. If you find yourself nudging up the TV volume, asking "what?" in conversations, or feeling lost in noisy environments, you are likely the perfect candidate.
These devices offer a phenomenal, accessible, and empowering entry point to regaining clarity. But if your hearing loss feels severe, or is accompanied by other issues like tinnitus or dizziness, your first call should always be to a hearing health professional.
What’s the Difference Between FDA Approved, Cleared, and Registered?
These terms are not interchangeable; they represent different levels of FDA oversight.
- FDA Approved: The most rigorous review, reserved for high-risk, novel devices like cochlear implants.
- FDA Cleared: Applies to most prescription hearing aids. It means the FDA has determined the device is "substantially equivalent" to a device already legally on the market.
- FDA Registered: The category for most OTC hearing aids. The manufacturer has listed its device with the FDA and self-certified that it meets all federal safety and performance standards.
Can I Really Buy a Legitimate, High-Quality Hearing Aid Online?
Absolutely. The ability to purchase directly from trusted brands online is one of the greatest advancements in hearing health accessibility. Reputable companies like HearDirectClub offer FDA-registered devices that meet the highest standards, delivered right to your door.
The key is to choose a transparent company. Ensure the seller explicitly states their products are FDA-compliant medical devices and offers robust customer support and a strong satisfaction guarantee.
Are you ready to turn the volume up on your life? The HearDirectClub Nova hearing aid is an FDA-registered, Class I device engineered to bring the world back into sharp, vibrant focus. With our 100-day risk-free trial, you have nothing to lose and a world of rich, beautiful sound to regain.





Leave a comment
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.